WHAT ON EARTH ARE HALOGENS?
Halogens are elements from group 7. This means they all only need 1 electron to get a full outer shell. They will try to react with other elements to get this extra electron. One important thing to remember, Halogens are diatomic, they will pair up with each other like oxygen does. Chlorine is Cl2 , Bromine Br2 etc.
WHICH ARE MORE REACTIVE?
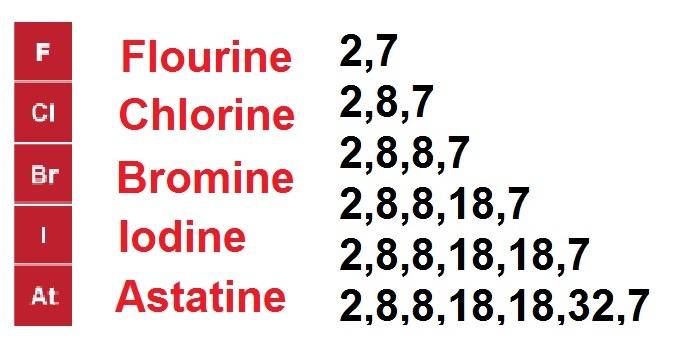
Unlike the alkali metals, the halogens are more reactive as you go up, Fluorine being the most reactive. This is because as you have more and more shells of electrons, the outer shell is further from the nucleus, and has a weaker pull on electrons, meaning larger atoms find it much harder to react. On the flip side Fluorine only has a few electrons, so has a much stronger pull on electrons, allowing it to react more easily.
DISPLACEMENT REACTIONS
Hopefully you know about displacement reactions, if not listen up.
Displacement reactions are when a more reactive element displaces a less reactive element from it’s compound. If you remember the reactivity series then you will know that putting an iron nail into copper sulphate solution leads to a displacement reaction, the iron replaces the copper to form iron (ferrous) sulphate. The same type of reaction can be used to show the reactivity of the halogens.
A simple example of this is mixing sodium bromide solution with chlorine gas. Soon the solution turns brown; the bromine has been displaced by the more reactive chlorine to form Sodium Chloride (Table Salt).
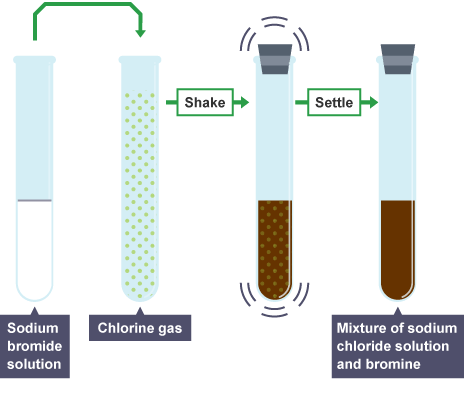
These displacement reactions are redox reactions because the chlorine gains an electron (reduction) and the bromine loses an electron (oxidation).
WHAT DO HALOGENS EVEN LOOK LIKE?
THAT'S ALL YOU NEED TO KNOW ABOUT HALOGENS!
I hope this was useful, if not let me know in the comments! If you want me to explain a topic for you let me know in the comments.