What are carbohydrates?
Carbohydrates have the general formula Cx(H2O)y. Carbohydrates that you’re likely to be familiar with are sugars, starch and cellulose.
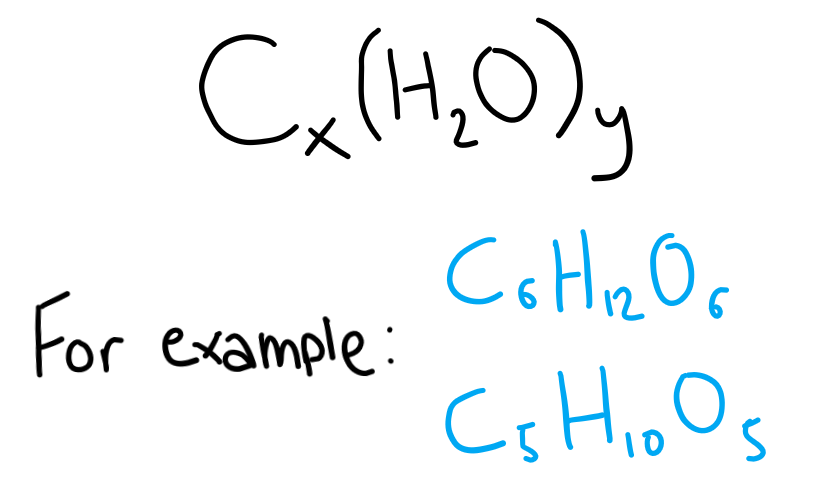
Monosaccharides vs Polysaccharides
Monosaccharides are carbohydrates with between 3 and 6 carbons.
Polysaccharides are carbohydrates with more than 6 carbons. These are usually polymers of monosaccharides.
Sugars 🍩🍫🍬🍭
Sugars are monosaccharides, and have between 3-6 carbons. They come in both straight chain and cyclic form (more on this later).
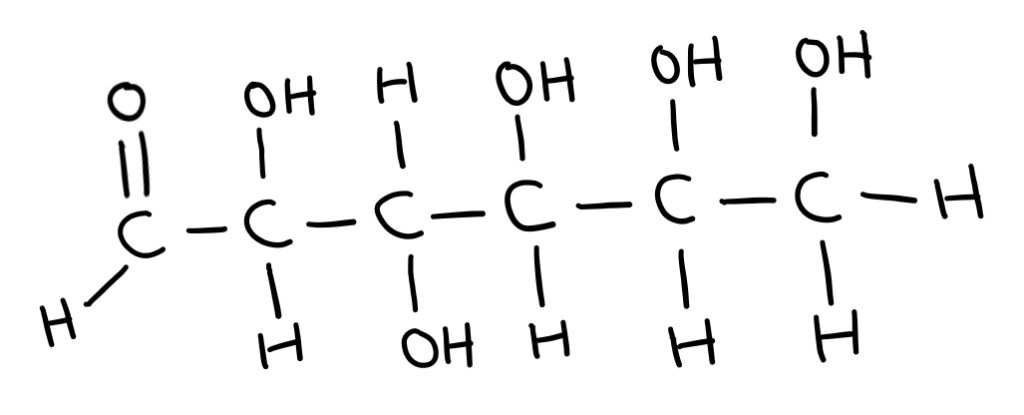
In straight chain form, sugars can either have an aldehyde or a ketone group.
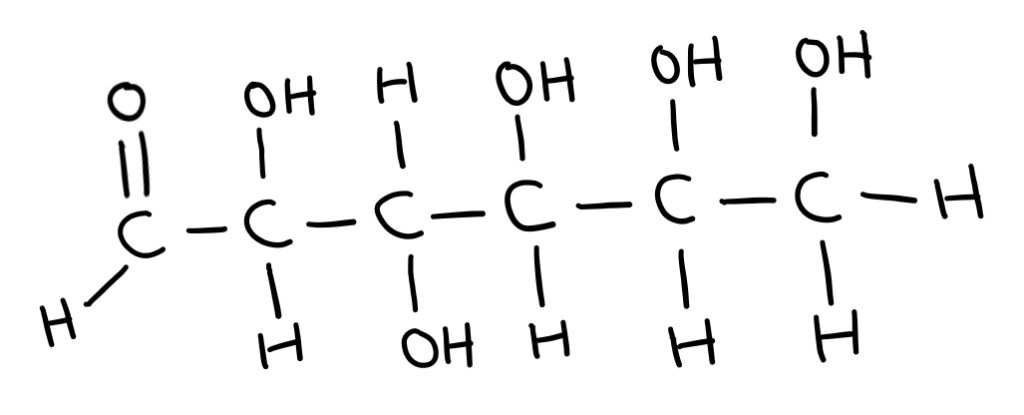
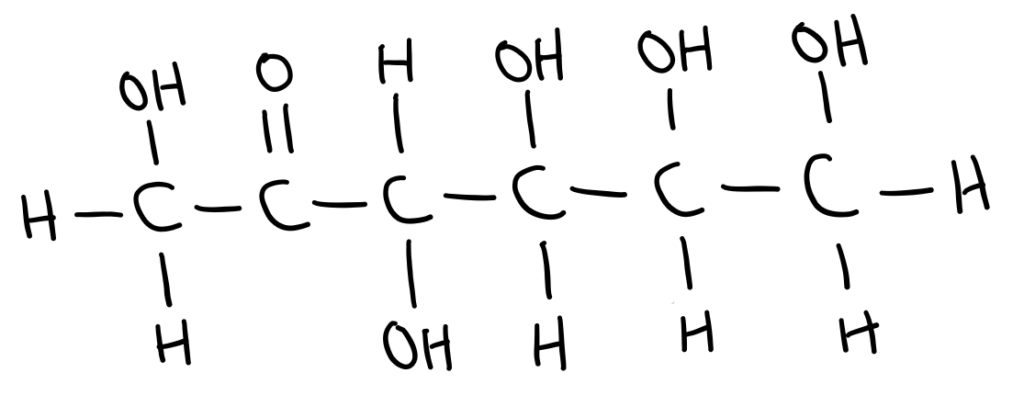
Sugars with an aldehyde group are called aldoses and sugars with ketone groups are called ketoses.
Sugars are also named and classified depending on the number of carbons they have.
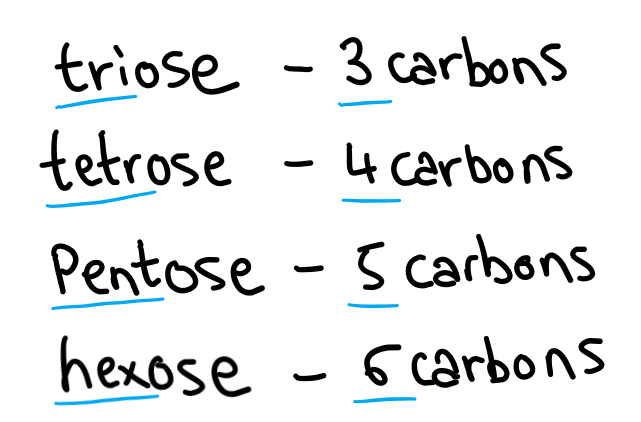
When you combine these together you can make cool words like aldohexose and ketohexose.
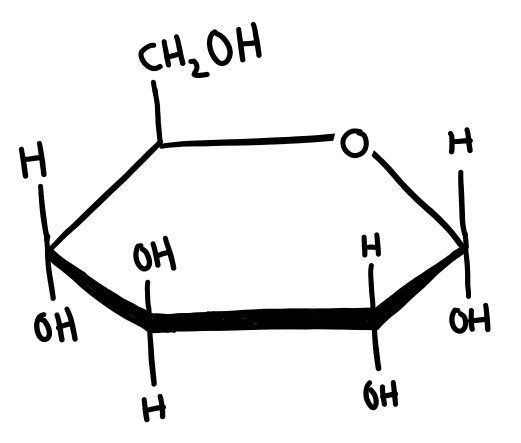
Cyclisation of sugars
In solution, some monosaccharides (pentoses and hexoses) form cyclic compounds. The carbonyl (aldehyde or ketone) group reacts with a hydroxyl group to form the ring structure.
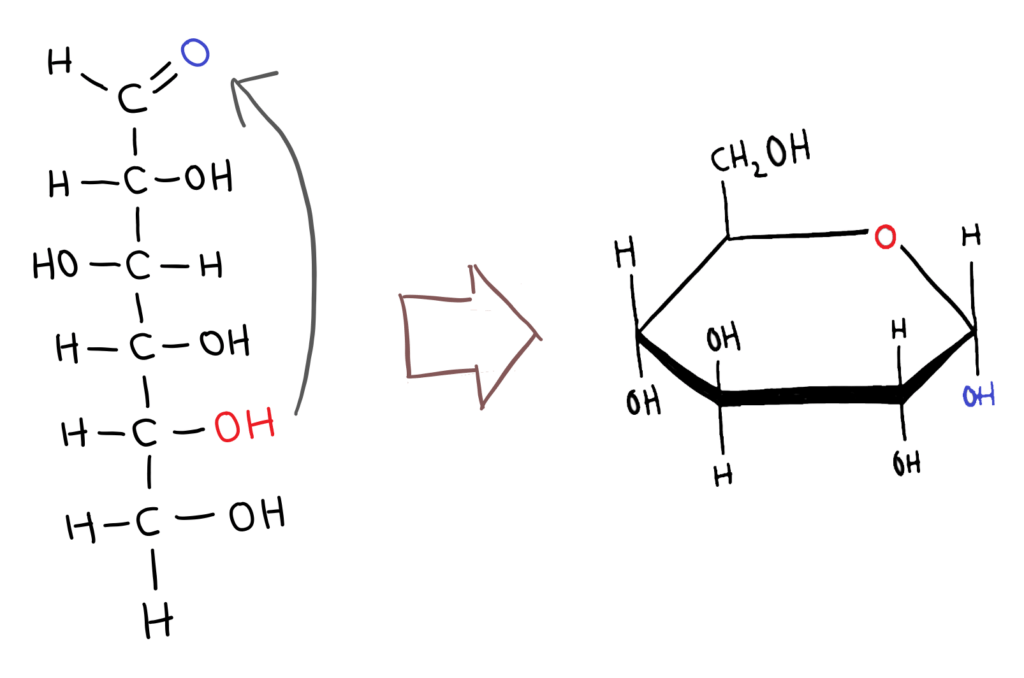
The mechanism of the cyclisation of sugars is hard to visualise but I’m working on it ☺😰
Trioses and tetroses don’t cyclise because the bond angle between the carbons in the ring are very small which leads to a large amount of ring strain.
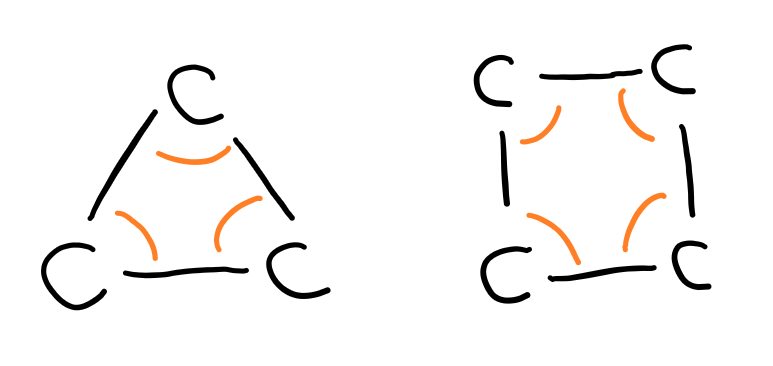
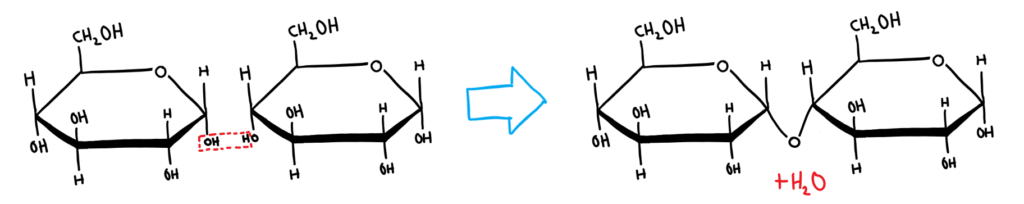
Disaccharides
Disaccharides are formed from two monosaccharides that react together in a condensation reaction. This reaction forms a glycosidic (ether) linkage. This linkage is called a 1,4 linkage because the bond forms between carbon 1 and 4 in the monosaccharides.
Polysaccharides
Polysaccharides are basically just monosaccharides that bond together in the same way as disaccharides (above) to form long chains. I spent a considerable amount of time making the diagram below to illustrate the length of these chains (please don’t roast me or i will be very sad)
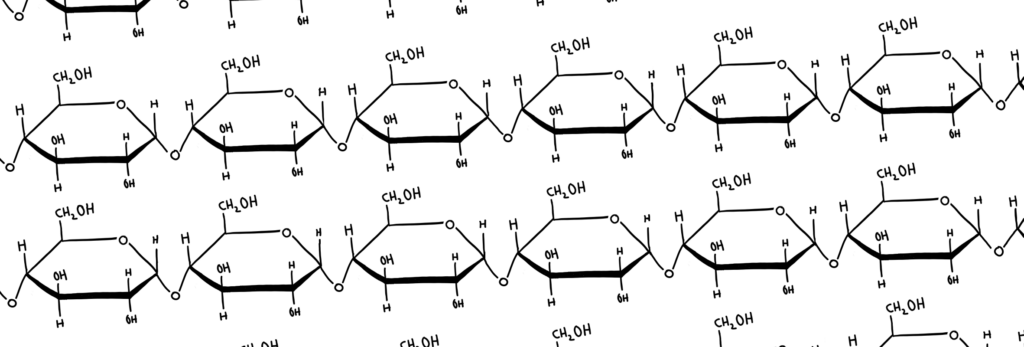
Some examples of polysaccharides include:
- Starch (Amylose and Amylopectin)
- An energy source found in foods
- Glycogen
- Used as energy storage in humans
- Cellulose
- Used in the cell walls of plants to provide structure.
The way polysaccharides such as starch, glycogen and cellulose differ is discussed in B.10 – Stereochemistry in Biomolecules because it has to do with different isomers of the monosaccharides.