B.9 - Pigments
What are pigments?
Pigments are biological molecules that are coloured compounds produced in metabolic reactions. The difference between a pigment and a dye? A dye is synthetic and a dye is biological.
What causes pigments to be coloured?
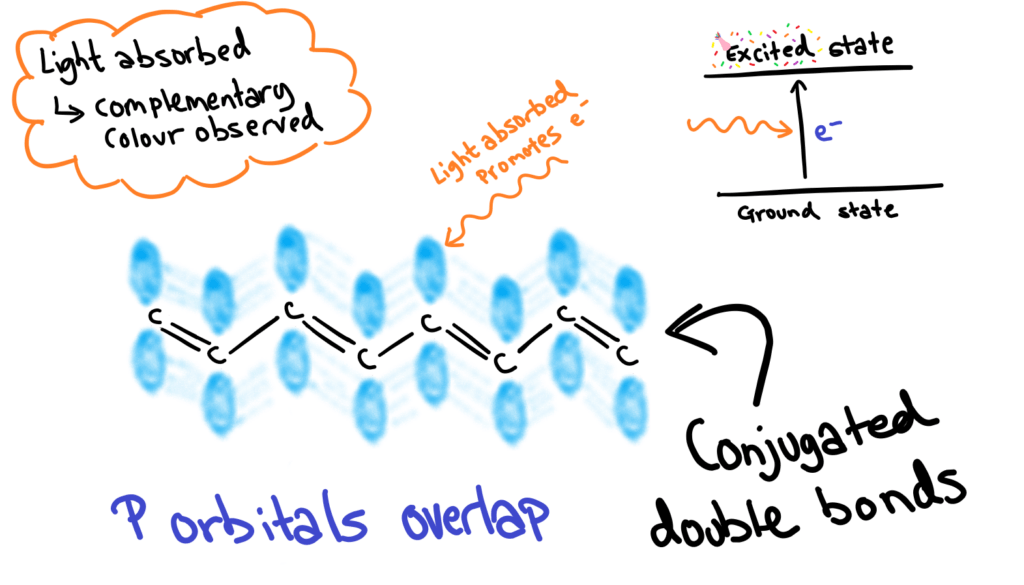
Pigments are usually large organic molecules. Their colour comes from light absorbed by conjugated double bond systems within the molecule. What is conjugation? It’s when you have alternating double and single bonds between carbons in an organic molecule.
The p-orbitals of the conjugated double bonds overlap slightly, and the electrons in these orbitals absorb light, and the complementary colour is observed.
REMEMBER: The longer the conjugation, the longer the wavelength absorbed
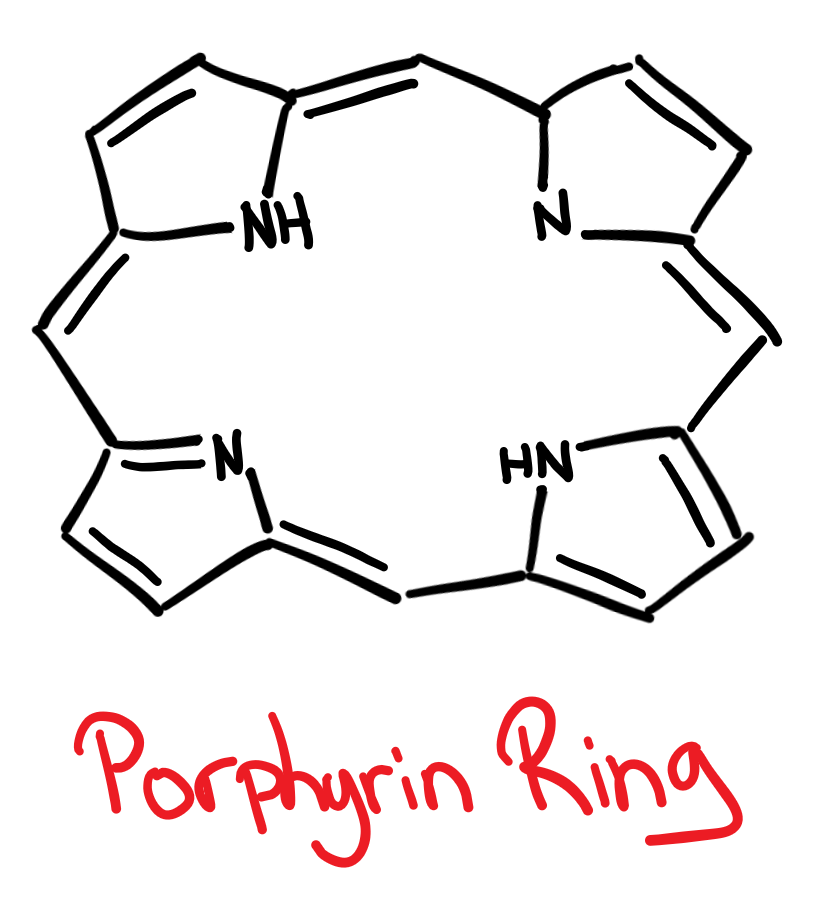
Porphyrin Pigments
Porphyrins are a class of pigments that all contain a porphyrin ring, which is highly conjugated. The porphyrin ring is a:
macrocyclic ligand
- macro=big
- cyclic=circle boi
- ligand= forms coordinate bonds
polydentate ligand
- poly= more than 1
- dentate= teeth? bitey bitey.
- ligand= forms coordinate bonds
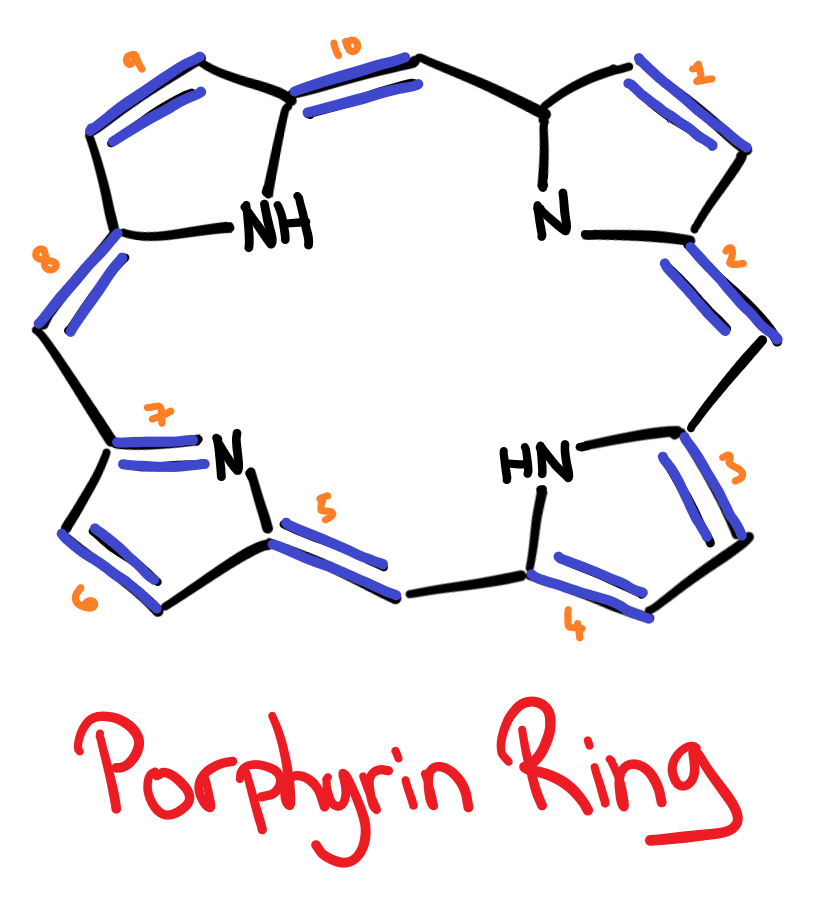
The nitrogen atoms in the porphyrin ring allows the ring to form 4 dative/coordinate bonds. Because of this, porphyrin pigments are always found as chelates, the 4 nitrogens are bound to a central metal atom.
Some examples of porphyrin pigments are:
- Chlorophyll(s) (Magnesium)
- Cytochromes (iron)
- Heamoglobin
- Myoglobin
In cytochromes, the iron is able to form 6 coordinate bonds, 4 of which are formed between the iron and the porphyrin ring.
In haemoglobin the iron also binds to a protein chain and an sometimes an oxygen molecule.
In oxyhaemoglobin, the iron atom is in the 3+ oxidation state and in deoxyhaemoglobin, it’s in the 2+ oxidation state.
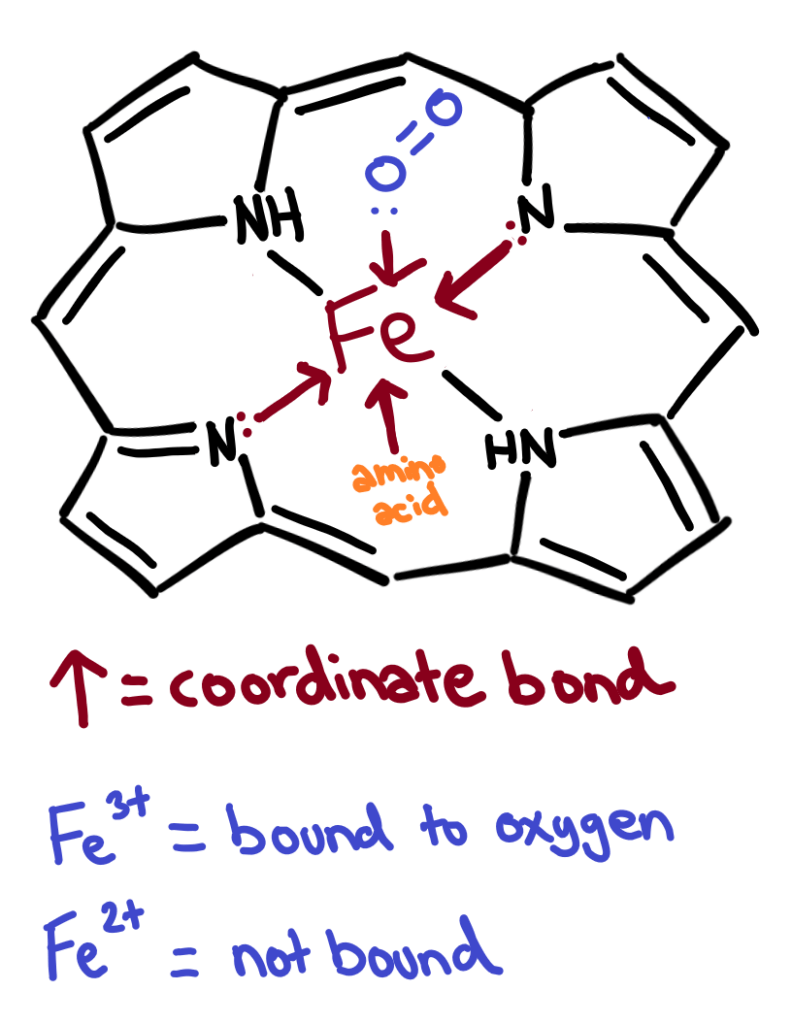
Haemoglobin in more detail 😎
Adult haemoglobin contains 4 heme groups each with a protein chain attatched. Each heme group can bind one oxygen molecule, but the bond to the oxygen from the iron atom is weak, allowing the oxygen to be released when needed.
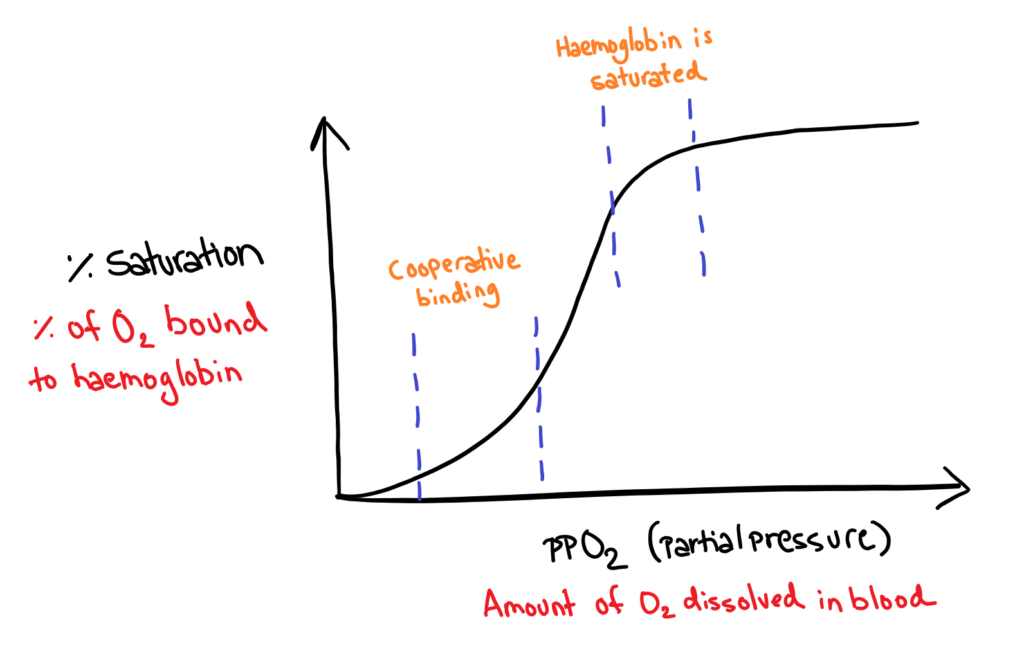
Oxygen dissociation
The binding of oxygen to heamoglobin is shown in a graph of %saturation in haemoglobin against partial pressure of O2 in the blood stream.
This graph forms a sigmoidal shape for two reasons.
- In the beginning, binding of oxygen is difficult. But when the first oxygen binds, there is a conformational change in the protein subunits that make binding more oxygen easier. This is called cooperative binding.
- After a lot of oxygen is bound, the haemoglobin is saturated, and binding more oxygen is therefore difficult.
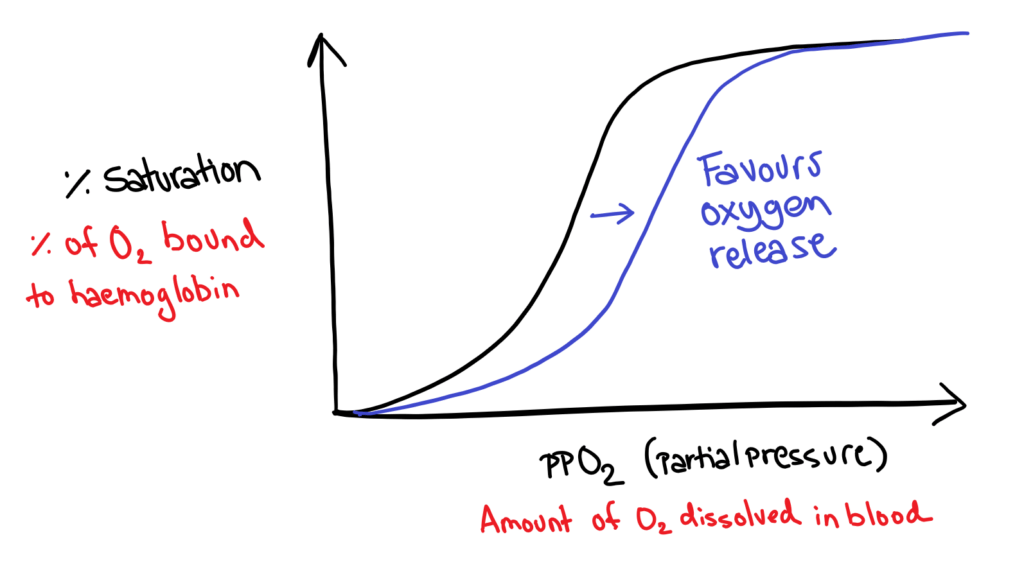
Factors that affect oxygen binding
In order to favour oxygen release (as opposed to binding), the graph needs to be shifted right (binding must be harder). This happens when:
- Increased CO2 levels
- Increased acidity (due to CO2)
- Increased temperature
These are all things that happen during respiration (yay😊)
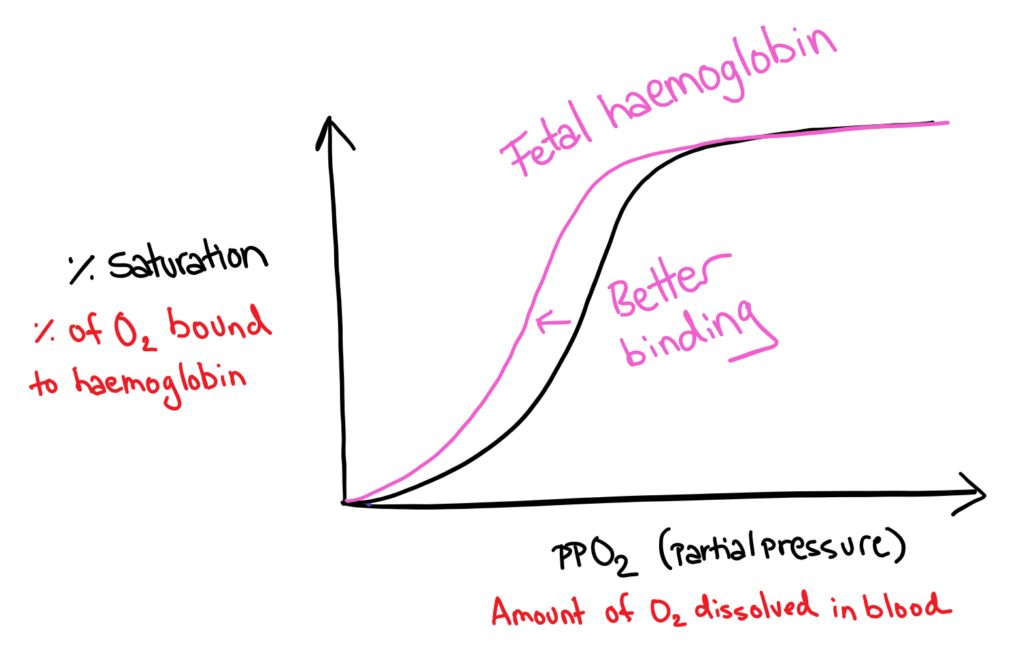
Fetal Haemoglobin
When a fetus is growing inside the mother, it obviously cannot use it’s lungs to get oxygen from the air, it must take oxygen from the mother’s blood. In order to do this, fetal haemoglobin must have a higher affinity for oxygen than the mother’s adult haemoglobin.
Fetal haemoglobin has a higher affinity for oxygen because it has two different protein chains to adult haemoglobin that favour oxygen binding; fetal haemoglobin has 2 gamma units instead of 2 beta units.
Carbon monoxide poisoning 😢
Carbon monoxide poisoning occurs when carbon monoxide binds to haemoglobin. Haemoglobin’s affinity for carbon monoxide is about 200 times more than that of oxygen because carbon monoxide is a stronger lewis base.
Basically, if carbon monoxide binds to haemoglobin, it’s never coming off. This of course renders the haemoglobin useless and if you breathe in enough carbon monoxide, you will unfortunately die from a lack of oxygen.
Anthrocyanins
Finally, a new pigment 😁. Anthrocyanins are aromatic, water soluble pigments that are found in plants. Their colour depends on the pH of the environment they’re in. Under different pH conditions, anthrocyanins will react with free H⁺ or OH⁻ ions. This reaction will result in a ‘chain reaction’ that changes the conjugation of molecule, therefore changing it’s colour.
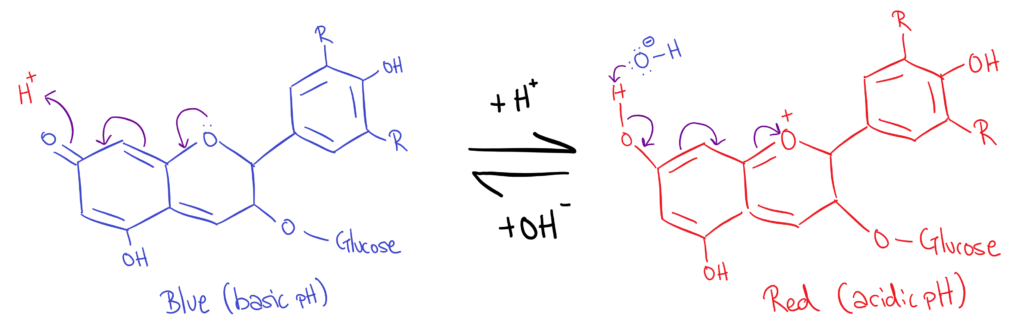
The fact that anthrocyanins change colour based on pH makes them good indicators!
HOW TO BREAK ANTHROCYANINS (enhanced interrogation techniques don’t tell the government)
- Anthrocyanins also form complexes with metal ions which destroy their colour
- Will break down at high temperature
- Rings break which also breaks conjugation
- And also at extreme pHs
- H and OH⁻ ions react with like everything and destroy the colour
Chlorophylls and the Carotenoids
Chlorophylls and carotenoids are large non-polar (lipid soluble) pigments that are found in plants and some bacteria that are involved in photosynthesis (ask the biologists for more deets).
The Chlorophylls
Shock and horror there are more than just 1 chlorophyll. Chlorophyll A & B absorb different wavelengths of light due to different degrees conjugations, allowing photosynthesis to use more energy from light to do important things like make glucose.
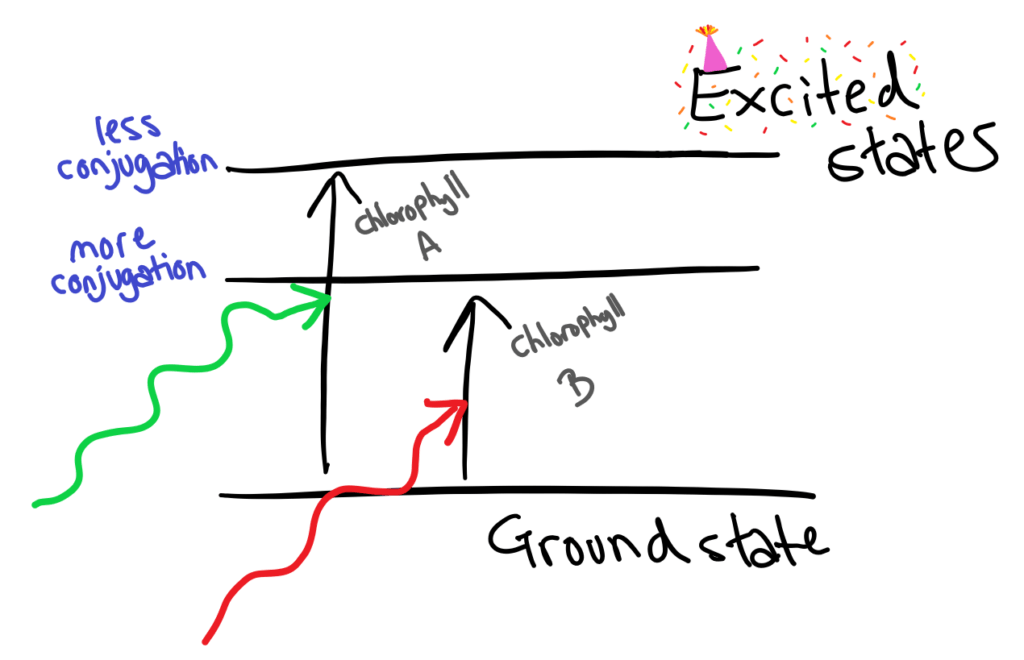
Stability
- Destroyed by heat
- Magnesium ion is lost from porphyrin ring
- Damaged more easily than carotenoids
- Easier to lose Mg ion than break carotenoid bonds
- Destroyed by Low pH
- Hydrogen ions react with nitrogens in the porphyrin ring and kick out the magnesium ion.
- Double bonds destroyed by light
- Double bonds are oxidised as catalysed by light
- Similar to oxidative rancidity of lipids!
Carotenoids
Carotenoids are another group of pigments found in plants that are involved in absorbing light for photosynthesis. The two carotenoids IB is interested in are α and β-carotene.
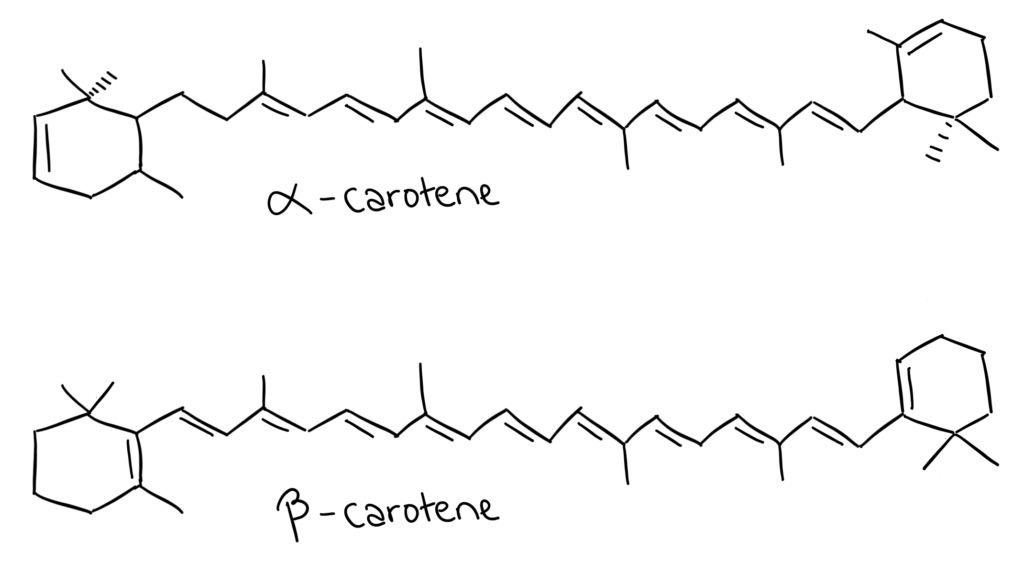
- α-carotene is less conjugated
- Absorbs a higher wavelength
- β-carotene is more conjugated
- Absorbs a lower wavelength
Stability
- Destroyed by heat
- Heat breaks double bonds and therefore conjugation 😢
- Destroyed by light
- Light oxidises the double bonds in the same way as chlorophyll
Thin Layer Chromatography (TLC)
It’s chromatography. On a thin layer. Of glass.
Go figure 🙂
Just kidding I’ll do this tomorrow bc it’s 12am right now and I’m a delicate little flower who needs sleep
Thank you so much! Can you please add the notes on TLC?